
The Multiple Functions of Cysteine-String Protein Analyzed at Drosophila Nerve Terminals | Journal of Neuroscience
Cysteine String Protein Limits Expression of the Large Conductance, Calcium-Activated K+ (BK) Channel | PLOS ONE

Neurons Export Extracellular Vesicles Enriched in Cysteine String Protein and Misfolded Protein Cargo | Scientific Reports

RCSB PDB - 2N04: Solution Structure of the phosphorylated N-terminal region of Human Cysteine String Protein (CSP)

The Role of Cysteine String Protein α Phosphorylation at Serine 10 and 34 by Protein Kinase Cγ for Presynaptic Maintenance | Journal of Neuroscience
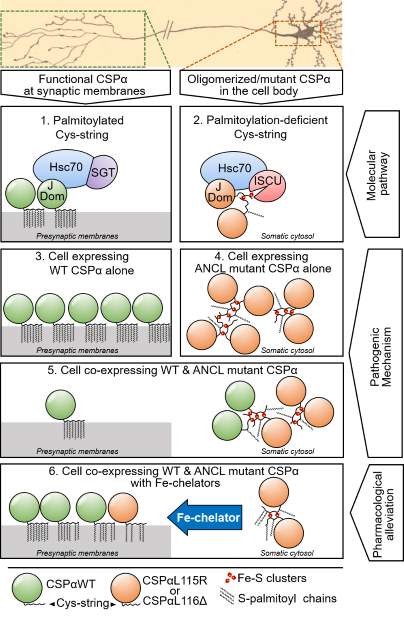
An Unusual Mechanism of Pathogenic Aggregation of Cysteine String Protein-α (CSPα) | Helen & Robert Appel Alzheimer's Disease Research Institute
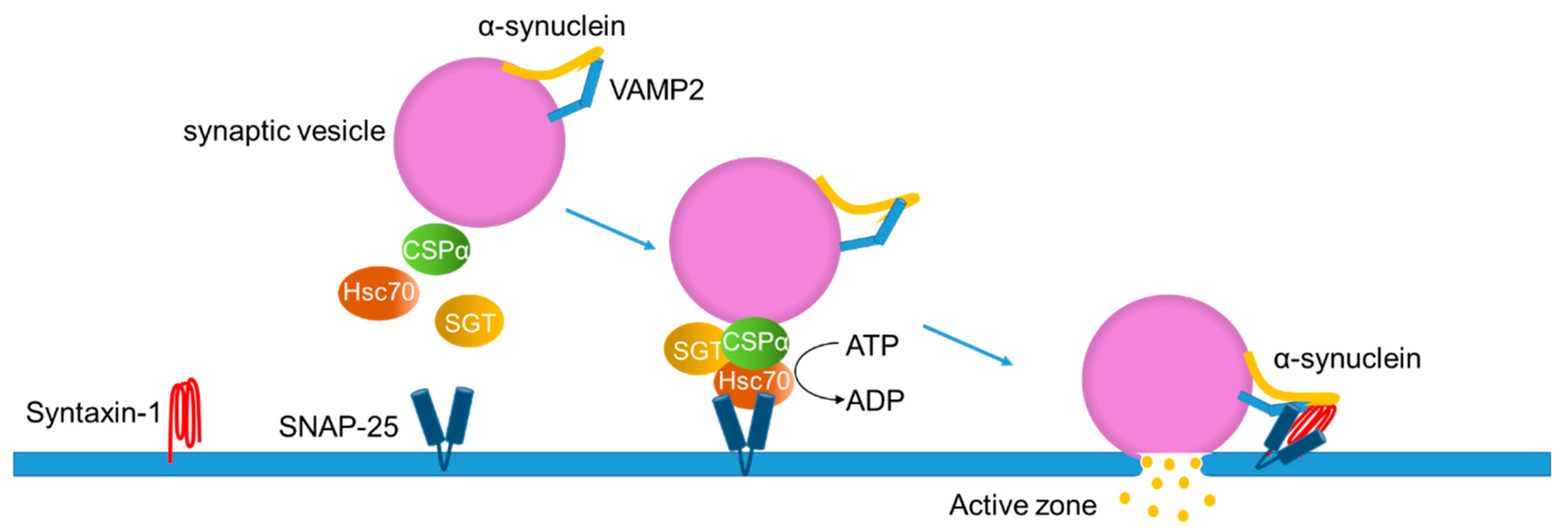
IJMS | Free Full-Text | Lipids: Key Players That Modulate α-Synuclein Toxicity and Neurodegeneration in Parkinson's Disease

Aggregation of mutant cysteine string protein-α via Fe–S cluster binding is mitigated by iron chelators | Nature Structural & Molecular Biology
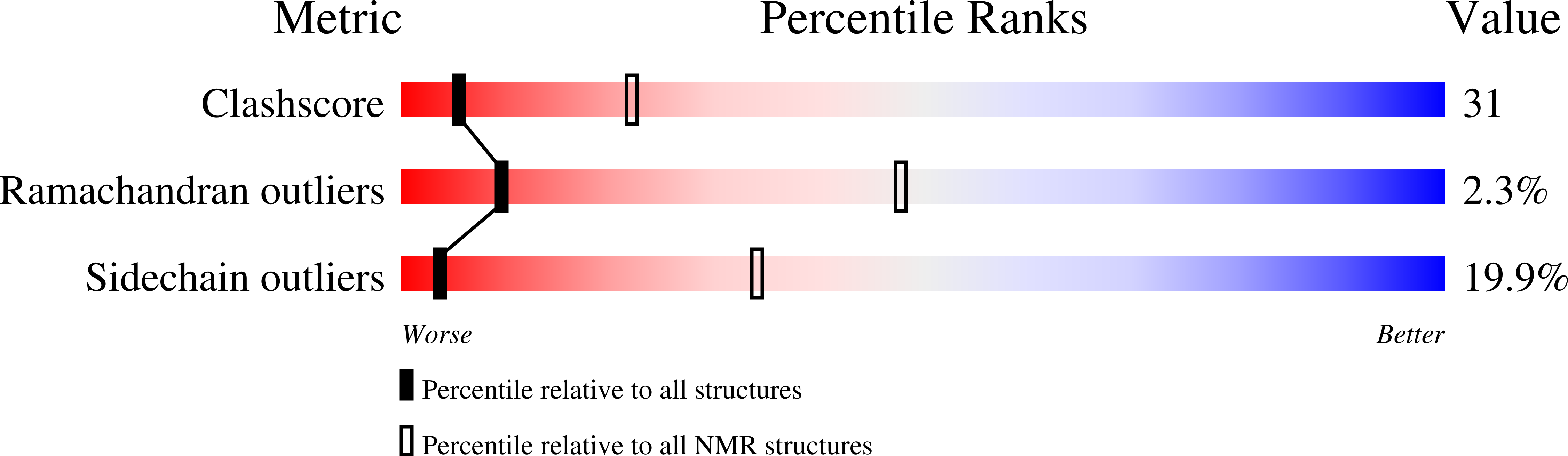
RCSB PDB - 2N04: Solution Structure of the phosphorylated N-terminal region of Human Cysteine String Protein (CSP)

Aggregation of mutant cysteine string protein-α via Fe–S cluster binding is mitigated by iron chelators | Nature Structural & Molecular Biology
Tying Everything Together: The Multiple Roles of Cysteine String Protein (CSP) in Regulated Exocytosis
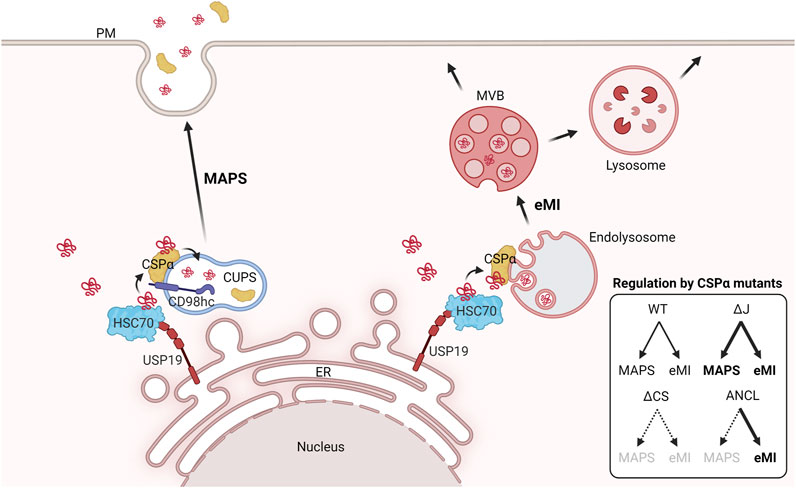
Frontiers | Safeguarding Lysosomal Homeostasis by DNAJC5/CSPα-Mediated Unconventional Protein Secretion and Endosomal Microautophagy

Aggregation of mutant cysteine string protein-α via Fe–S cluster binding is mitigated by iron chelators | Nature Structural & Molecular Biology











